- News
QuTEM Collaborates with Boston University to Bring Advanced Quality Control Analysis to American Gene Therapy Developers
Stockholm, MARCH 10th, 2026
QuTEM and Boston University collaborate to provide localized, high-speed AAV analysis for North American gene therapy developers.
QuTEM AB, a world-leading provider of GMP-certified TEM-services and quality control analyses of nanoparticles, today announced the successful launch of its U.S. satellite laboratory operations in collaboration with The Boston University Cryogenic Electron Microscopy (BU CryoEM) Core Facility. Marking the milestone, QuTEM has successfully completed and delivered its first client project through this newly established transatlantic workflow.
The association allows US-based gene therapy developers to seamlessly access QuTEM’s premier analytical services and proprietary Gridsee software without the logistical complexities of shipping R&D-level samples internationally.
By establishing a physical footprint in the heart of the world’s premier biotech hub, QuTEM provides immediate, localized support to leading developers. This approach drastically compresses turnaround times by eliminating transatlantic shipping and potential customs delays. Furthermore, operating across two continents provides an operational redundancy and supports uninterrupted service for time-sensitive R&D programs.
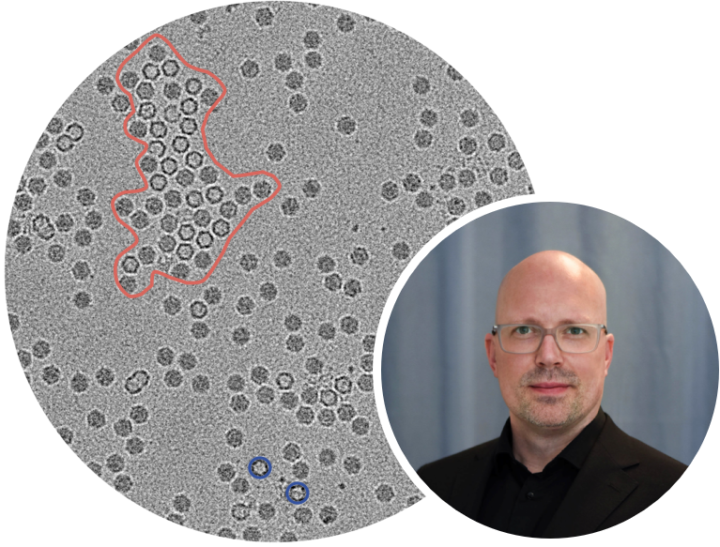
Through this expanded workflow, clients send their R&D-level Adeno-Associated Virus (AAV) samples directly to the Boston University facility. Operating under QuTEM’s standardized operating procedures (SOPs), Boston University’s TEM scientists perform the sample preparation and TEM imaging. The images are then securely transferred to QuTEM’s headquarters in Sweden, where experts utilize the Gridsee software to evaluate Critical Quality Attributes (CQAs), such as empty/full/intermediate ratios and aggregation, delivering comprehensive, objective quality control reports.
"Because the Boston University facility operates the exact same microscopy equipment as our primary laboratories in Stockholm, we eliminate the technical uncertainties typically associated with scaling up," said Josefina Nilsson, CEO at QuTEM AB. "Clients can accelerate their pipelines locally, completely confident that their R&D data will translate seamlessly as they transition to our European facilities for GMP compliance."
Boston University operates this partnership under its established fee-for-service model, providing QuTEM with highly efficient, dedicated hourly access to its Core Facilities. This allows QuTEM to expand its physical U.S. presence while maintaining end-to-end quality control over the analytical pipeline and data integrity.
Following the success of this initial project, the Boston University satellite lab is fully operational and actively accepting new U.S. clients. QuTEM is now positioned to rapidly expand its support for North American gene therapy developers, delivering AAV quality control with speed, security, and uncompromised precision.
More news
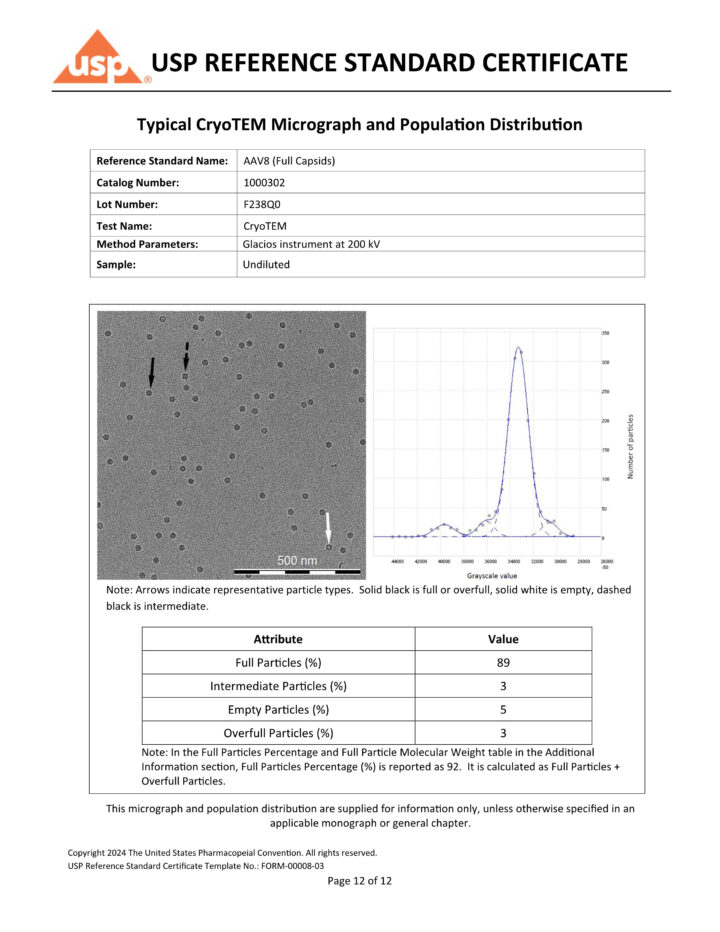
CryoTEM recognized as official analytical method in USP AAV8 reference standard certificates

New publication on AAV Characterization: The Problem of Partials